Research
All organisms need to adjust their behavior to adapt to a changing environment. Feeding behaviors, for example, depend upon both internal states (i.e. hunger) and external realities, (i.e. food availability). Stress influences food-based decision making and metabolic outcomes, and certain stigmatized bodily and behavioral responses to external and internal stressors can lead to disease states, such as anorexia nervosa or obesity. Our translational laboratory uses systems neuroscience tools to better understand the pathophysiology and biology that underlies the behavior related to these diseases in hopes of reducing the associated stigma. We are focused on how neuropeptides regulate specific circuits at the interface of stress and metabolism, with an interest in sex differences and behavior differences that result in outcomes across the weight spectrum related to psychiatric and medical illness. We concentrate on two neuropeptidergic systems: the metabolism-associated melanocortinergic system, and the stress-linked PACAP system. In rodent models we use a combination of behavior studies, electrophysiology, in vivo Ca2+ imaging, pharmacologic, optogenetic, and chemogenetic manipulations to interrogate how these neuropeptides regulate neural circuits at the interface of stress and metabolism. In collaboration with clinical researchers, we work to apply our findings to inform investigations into human behavior using molecular, genetic, and qualitative approaches.
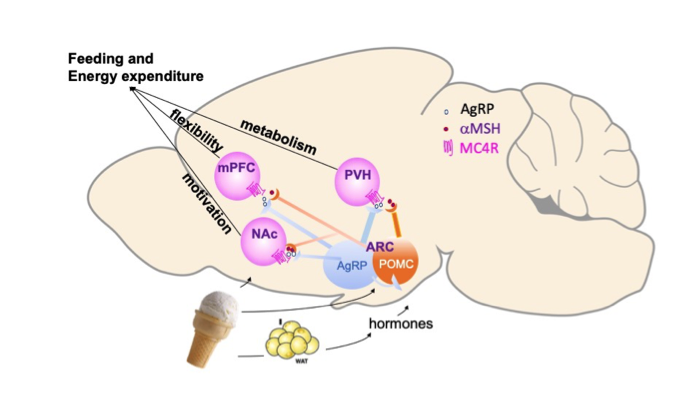
Current projects in the lab include:
- What is the role of the melanocortin-4 receptor in the medial prefrontal cortex in cognitive flexibility. Is it specific to food-related decision making? How do neurocircuit responses of males and females in different states lead to different body weight and food intake related outcomes?
- Does metabolic stress transmit a PACAPergic signal, and how is this different between males and females? How does this affect the reproductive axis?
- How does dietary manipulation affect behavioral outcomes?
- Can we model (and ameliorate) cognitive symptoms in the Prader Willi Syndrome mouse model of early life obesity and intellectual disability?
- How does early life food insecurity disrupt the coordination of stress and metabolic neuroendocrine signalling critical to healthy development, physiology, and behavior?